


On 20 February 2025, CMA Testing was invited to participate in the online live broadcast titled “Set Sail, Set Sail from the Harbor” organized by Inner Mountain, DHGroup and HKTDC. The event aims to advocate for Hong Kong as an international free port and to highlight the benefits and opportunities available for Chinese domestic products seeking to go overseas. As one of the key partners of the organiser, CMA Testing was invited to participate in this event to showcase our testing compliance services, CMA+ product development and commercialisation services, and WINGS Future’s services to assist the export of high-quality agricultural products from the Mainland.

On December 16, 2024, the European Council officially adopted the Packaging and Packaging Waste Regulation (EU) 2025/40 (Packaging and Packaging Waste Regulation, PPWR), and it was officially signed by the European Parliament and Council on December 19. On January 22, 2025, the EU officially published PPWR in its Official Journal, repealing Packaging and Packaging Waste Directive 94/62/EC. The PPWR will enter into force on February 11, 2025, and be implemented starting from August 12, 2026 (unless otherwise specified).
Scope of PPWR:
The regulation is applies to all packaging, regardless of the material used, and to all packaging waste, whether such packaging is used in or such packaging waste originates from industry, other manufacturing, retail or distribution, offices, services or households.
Key Provisions of PPWR:
- Requirements for the entire life-cycle of packaging as regards environmental sustainability and labelling
- Extended producer responsibility
- Prevention requirements for packaging waste
Core Requirements of PPWR:
- Requirements for substances in packaging
| Packaging Type | Requirements |
| All Packaging | Lead (Pb), Cadmium (Cd), Mercury (Hg), and Hexavalent Chromium (Cr6+) ,the sum of the concentrations ≤ 100mg/kg |
| Food-contact packaging | Any individual target PFAS substance content < 25ppb (polymeric PFAS excluded from quantification); The sum of PFAS measured as the sum of targeted PFAS analysis, where applicable with prior degradation of precursors < 250ppb (polymeric PFAS excluded from quantification); Total PFAS content < 50ppm (including polymer PFAS). If total fluorine content exceeds 50mg/kg, proof must be provided indicating the amount of fluorine present as PFAS or non-PFAS fluorine. |
- Recyclability Requirements for Packaging
All packaging placed on the EU market must be recyclable.
Packaging must be designed for material recycling and capable of replacing raw materials, with separate collection and classification when it becomes waste.
Starting from January 1, 2030, or 24 months after the enactment of the authorization act (whichever is later), only packaging with a recyclability performance grade of A, B, or C, as specified in Annex II Table 3 of regulation (EU) 2025/40, can be placed on the EU market. From January 1, 2038, only packaging meeting the recyclability performance grade of A or B in Annex II Table 3 can be placed on the EU market.
- Minimum Recycled Content Requirements for Plastic Packaging
| Packaging Type | Target | |
| By 2030 | By 2040 | |
| Contact-sensitive packaging made from polyethylene terephthalate (PET) (except single-use plastic beverage bottles) | 30% | 50% |
| Contact-sensitive packaging made from plastic materials other than PET (except single-use plastic beverage bottles) | 10% | 25% |
| Single-use plastic beverage bottles | 30% | 65% |
| Other plastic packaging | 35% | 65% |
- Packaging Minimization
By 1 January 2030, the manufacturer or importer shall ensure that the packaging placed on the market is designed so that its weight and volume is reduced to the minimum necessary to ensure its functionality, taking account of the shape and material from which the packaging is made.
- Labeling Requirements of PPWR:
From 12 August 2028 or 24 months from the date of entry into force of the implementing acts adopted, whichever is the latest, packaging placed on the market shall be marked with a harmonised label containing information on its material composition in order to facilitate consumer sorting.
From 12 February 2029 or 30 months from the date of entry into force of the implementing act adopted, whichever is the latest, shall bear a label informing users that the packaging is reusable. Information on reusability and collection points can be provided through QR codes or other data carriers.
From 12 August 2028 or 24 months from the date of entry into force of the implementing act adopted, whichever is the latest, plastic packaging with a recycled material content label must comply with the implementation act.
- Ban on Single-Use Plastic Packaging:
From January 1, 2030, the following single-use plastic packaging will be prohibited from being placed on the market:
- Single-use plastic packaging for portioning;
- Single-use plastic packaging for pre-packaged fresh fruits and vegetables (up to 1.5 kg);
- Hotel and restaurant takeout food and beverage packaging;
- Single-serving condiment, sauce, cream, and sugar packets;
- Small single-use cosmetics and toiletries in the hospitality industry;
- Very lightweight plastic carrier bags.

On December 19, 2024, the EU adopted a proposal to ban bisphenol A (BPA) in food contact materials and published (EU) 2024/3190 on December 31, 2024. This regulation repeals (EU) 2018/213 and amends the plastic food contact materials regulation (EU) 10/2011. The new regulation will officially come into effect on January 20, 2025.
Key Amendments:
- Ban on Bisphenol A (BPA) and Its Salts: Prohibited in adhesives, rubber, ion exchange resins, plastics, printing inks, silicone, varnishes, coatings, and other materials and products, unless they meet the specific use conditions outlined in Annex II.
- Prohibition of BPA Residues: Food contact materials made from other bisphenols or bisphenol derivatives must not contain residual BPA.
- Restriction on Other Harmful Bisphenols: Non-BPA harmful bisphenol substances and their derivatives may only be used with specific authorization.
- Declaration of Compliance Requirements: The specific category of bisphenols and their derivatives used must be declared.
- Amendments to (EU) 10/2011:
- Restricts the use of BPA and other harmful bisphenols, applying only to products that meet the exemptions specified in Annex II of (EU) 2024/3190.
- Removes two authorized substances from Table 1 of Annex I of (EU) 10/2011: BPA (CAS: 80-05-7) and BPS (CAS: 80-09-1).
Transition Arrangements:
- Single-use food contact final products made with BPA that comply with previous regulations but do not meet the new regulation:
- May continue to be sold until July 20, 2026.
- The following special products may be sold until January 20, 2028:
- Containers for preserving vegetables or fruits (excluding juices and similar products).
- Containers for preserving seafood.
- Products with BPA-based varnish or coating applied to the outer surface of metal.
- Reusable food contact final products made with BPA that comply with previous regulations but do not meet the new regulation:
- May be placed on the market for the first time until July 20, 2026, with stock sales allowed until January 20, 2029.
- Reusable products used in professional food production equipment that comply with previous regulations may be placed on the market for the first time until January 20, 2028, with stock sales allowed until January 20, 2029.

Recently, CMA Testing (Yu Biao) received the 2025 Compliance Antimicrobial Testing Laboratory Certification from the China Antibacterial Materials and Products Industry Association (CIAA). The antimicrobial test reports issued can now include the”CIAA Recommended Laboratory” logo!

Textiles CMA/CNAS Microbiological Accreditation
| Category | Regulation | Testing Items |
| Textiles | FZ/T 73023、 GB 20944 series | Antibacterial effectiveness ,Dissolution of Antimicrobial substances, Antibacterial (or bacteriostatic) |

On 21 January 2025, the European Chemicals Agency (ECHA) has added five new chemicals to the candidate list of substances of very high concern (SVHC). The Candidate List of SVHC now contains 247 entries for chemicals.
Entries added to the Candidate List 21 January 2025 and their properties:
| No. | Substance Name | EC No. | CAS No. | Reason for proposing | Use |
| 1 | 6-[(C10-C13)-alkyl-(branched, unsaturated)-2,5-dioxopyrrolidin-1-yl]hexanoic acid | 701-118-1 | 2156592-54-8 | Toxic for reproduction (Article 57c) | Lubricants, greases, release products and metal working fluid |
| 2 | O,O,O-triphenyl phosphorothioate | 209-909-9 | 597-82-0 | Persistent, bioaccumulative and toxic, PBT (Article 57d) | Lubricants and greases |
| 3 | Octamethyltrisiloxane | 4203-497-4 | 107-51-7 | Very persistent, very bioaccumulative, vPvB (Article 57e) | Washing and cleaning products, polishing and waxing products, as well as cosmetics and personal care products |
| 4 | Perfluamine | 206-420-2 | 338-83-0 | Very persistent, very bioaccumulative, vPvB (Article 57e) | Anti-corrosion transmission fluid, dielectric insulating fluid, electronic component and device leak detection fluid |
| 5 | Reaction mass of: triphenylthiophosphate and tertiary butylated phenyl derivatives | 421-820-9 | 192268-65-8 | Persistent, bioaccumulative and toxic, PBT (Article 57d) | Hydraulic oil, lubricants and greases |
In August 2024, ECHA released another substance for consultation, “Tris(4-nonylphenyl, branched) phosphite”. Regardless of the substance itself or when it contains ≥ 0.1% w/w of , “4-nonylphenol, branched and linear (4-NP) “, it has endocrine disrupting properties affecting the environment. Therefore, ECHA updated the “Tris(4-nonylphenyl, branched and linear) phosphite (TNPP) with ≥ 0.1% w/w of 4-nonylphenol, branched and linear (4-NP) “added in the 21st batch SVHC to “Tris(nonylphenyl, branched and linear) phosphite (TNPP) “
| Substance Name | EC No. | CAS No. | Reason for inclusion | Use | |
| Tris(4-nonylphenyl, branched and linear) phosphite | – | – | Endocrine disrupting properties (Article 57(f) – environment) | Polymers, adhesives, sealants and coatings | |
Under REACH, companies have legal obligations when their substance is included – either on its own, in mixtures or in articles – in the Candidate List.
If an article contains a Candidate List substance above a concentration of 0.1 % (weight by weight), suppliers have to give their customers and consumers information on how to use it safely. Consumers have the right to ask suppliers if the products they buy contain substances of very high concern. Importers and producers of articles have to notify ECHA if their article contains a Candidate List substance within six months from the date it has been included in the list (21 January 2025).
Under the Waste Framework Directive, companies also have to notify ECHA if the articles they produce contain substances of very high concern in a concentration above 0.1 % (weight by weight). This notification is published in ECHA’s database of substances of concern in products (SCIP).

The CMA has introduced a new special series titled “New Quality, New Vision”. Each episode features distinguished guests from various sectors who engage in-depth exchanges on how Hong Kong can develop a “Hong Kong version of new quality productivity” across all dimensions. The first episode was hosted by Dr. Wingco Lo Kam-wing, President of the CMA, and the Hon Jimmy Ng Wing-ka, Member (Industrial-Second) of the Legislative Council of the HKSAR, and featured an interview with Dr. the Hon Leung Chun-ying, Vice Chairman of the National Committee of the Chinese People’s Political Consultative Conference, discussing how Hong Kong can unleash “Alloy Productivity” with the mainland. As an entity, CMA Testing and CMA+ play a vital role in promoting the development of “Alloy Productivity”. In this latest episode, they introduce how one of our key partners, “Full of Hope”, exemplifies the effectiveness of “Alloy Productivity”.


Public access link:
Explore the CMA Channel for watching more content:
Facebook: https://www.facebook.com/HashtagCMA
YouTube: https://www.youtube.com/@HashtagCMA
Dear Customers and Partners,
Thank you for your continued support of our company. All of our staff is sincerely grateful for your patronage! Starting from February 9, 2025, CMA Testing Shanghai with CMA+ will be relocating to a new address (specific contact details will be provided below). During the relocation period, our business operations may be temporarily affected. We will do our utmost to minimize the impact on you. Should you have any questions or require further information, please feel free to contact us through the contact details provided below.
We look forward to continuing to serve you in our new environment and hope to continue to have your support. We apologize for any inconvenience that may be caused during the relocation period and appreciate your understanding and cooperation.
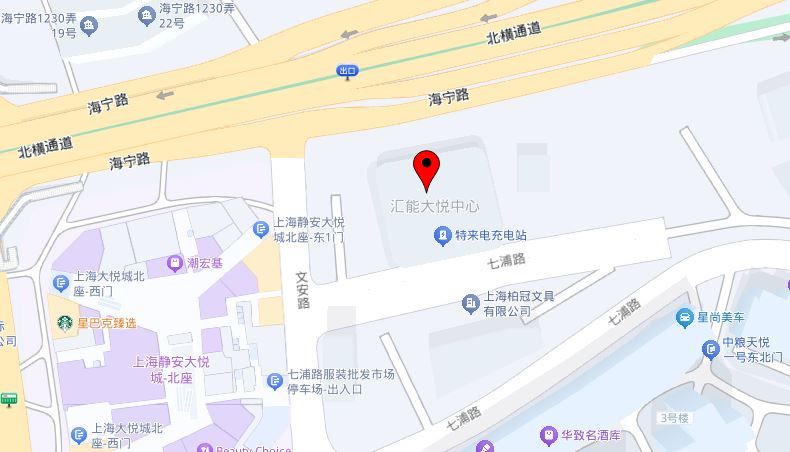
New Address:
F07, 9F, Huineng Center, No.1155, Haining Road, Jingan District, Shanghai
Tel: 021 – 64330500 (Remain Unchanged)
Email: info.sh@cmatesting.com.cn
Emergency contact: Stella ZHU 15000676303
CMA Testing and Certification Laboratories (Shanghai) Co., Ltd
with CMA+
Jan 2025

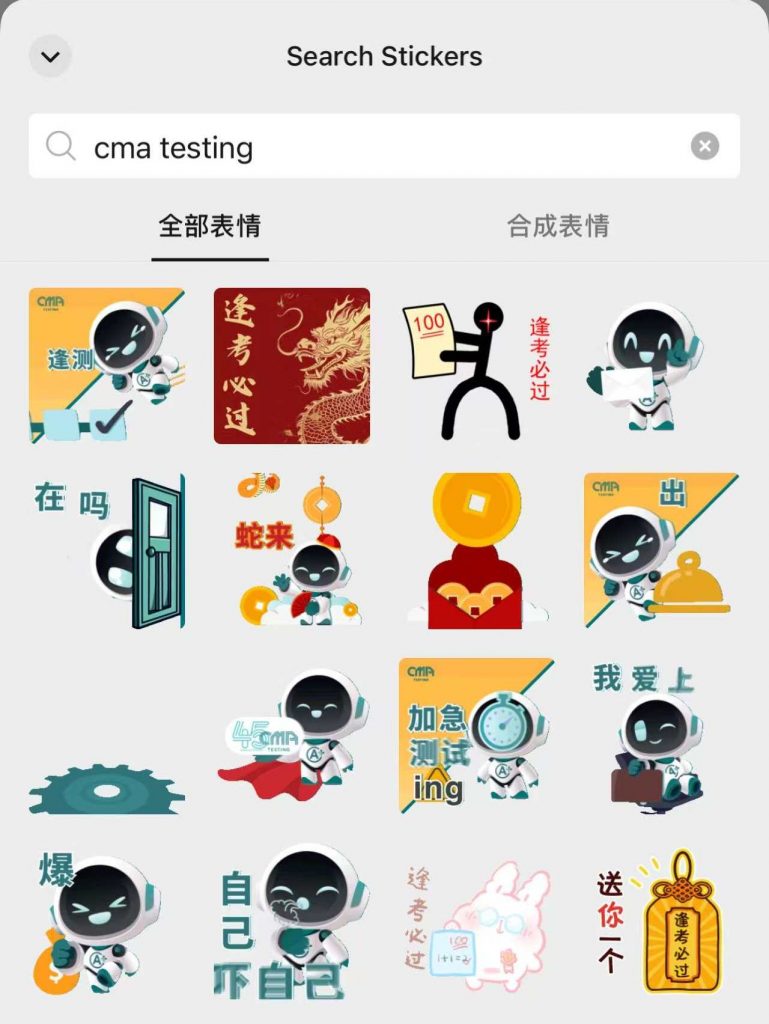
To celebrate the Chinese New Year, CMA+ have created a set of Dr A+ sticker packs for you to send well wishes to your family, friends, and clients. While nothing beats face-to-face engagement, it’s also hot and fun to share Chinese New Year stickers via Wechat! Download the Dr A+ sticker pack now to boost more innovation and fun during the festival!
How to download?
Method One:
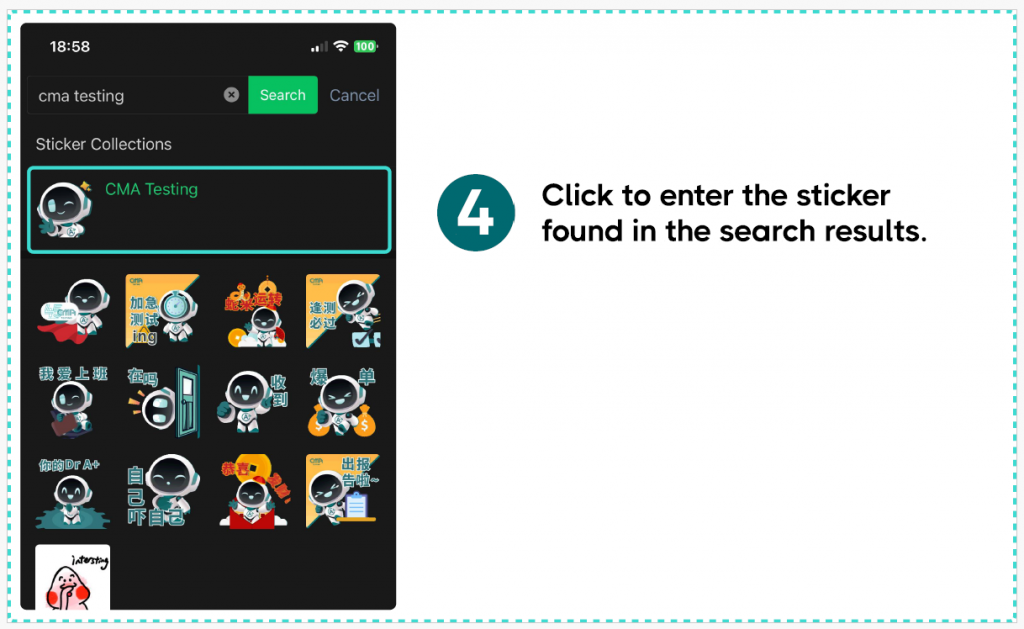

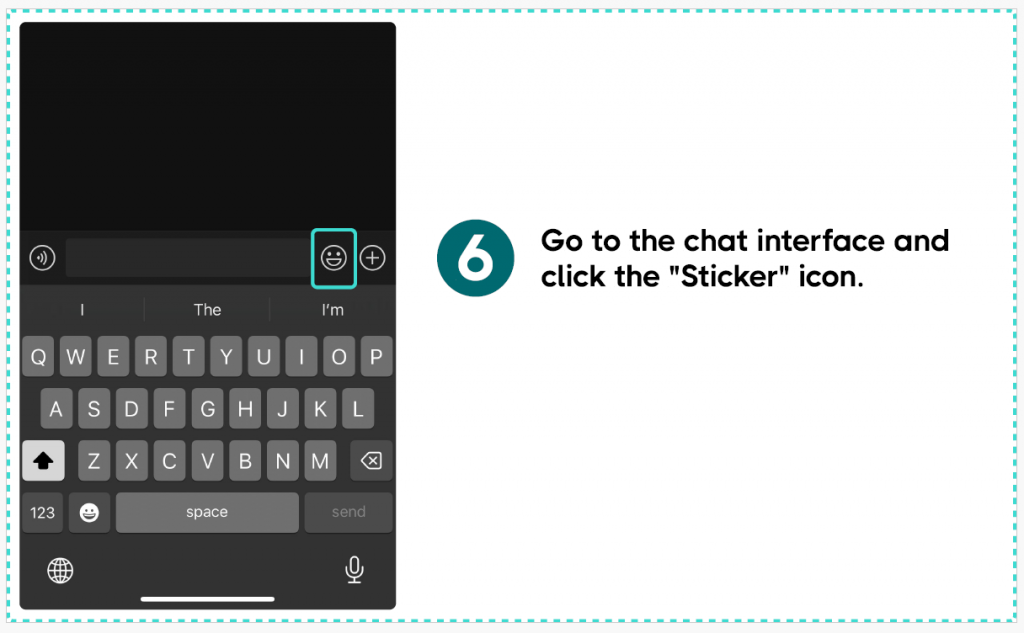

Method Two:
Acquisition Method 2
If you are using the Wechat International app, we recommend that you can select the emoji icon in any chat conversation and use its search function to search for “CMA Testing” to display it.

Greetings, everyone! We, at CMA Testing and CMA+, are pleased to take this opportunity to introduce our new team member to you, Dr A+! As a cutting-edge robot, Dr A+ is capable of working 24/7 tirelessly, embodying the characteristics of a workaholic, much like its pals at CMA+.
In the bustling world of technology and innovation, Dr A+ is dedicated to the pursuit of technology commercialisation, transforming aspirations into reality and making dynamic ideas available to all. With a strong commitment to education and support, Dr A+ diligently works to empower both individuals and organizations, ensuring that innovation transcends mere theory and turns research results into tangible tech solutions.
As an ideal inventor mentor, Dr A+ has a keen eye for potential and is dedicated to propelling your business forward. Whether you are launching a startup or aspiring entrepreneurs, Dr A+ is committed to catalyzing growth and success.
With a heart for collaboration and a mind for innovation, Dr A+ is not just a mascot but a symbol of hope and possibility in the ever-evolving landscape of technology.

